- Startseite
- butyl
- 𝘕,𝘕'-Diisopropyl-𝙊-𝙩𝙚𝙧𝙩-butyl isourea: an efficient reagent for 𝙩𝙚𝙧𝙩-butyl esters synthesis
𝘕,𝘕'-Diisopropyl-𝙊-𝙩𝙚𝙧𝙩-butyl isourea: an efficient reagent for 𝙩𝙚𝙧𝙩-butyl esters synthesis
4.9 (274) · € 19.99 · Auf Lager
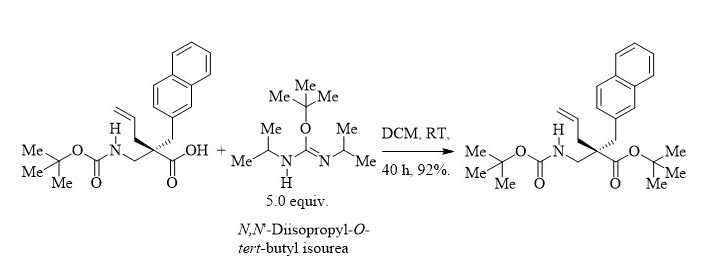
tert-Butyl esters are commonly used as protecting group of carboxylic acids in multi-step synthesis, especially with amino acids, peptides and natural products. [1] The importance of this relatively hindered protecting group is well established.

Efficient and Controllably Selective Preparation of Esters Using Uronium-Based Coupling Agents

𝘕,𝘕'-Diisopropyl-𝙊-𝙩𝙚𝙧𝙩-butyl isourea: an efficient reagent for 𝙩𝙚𝙧𝙩-butyl esters synthesis

Full article: Preparation and Applications of 4-Methoxybenzyl Esters in Organic Synthesis

𝘕,𝘕'-Diisopropyl-𝙊-𝙩𝙚𝙧𝙩-butyl isourea: an efficient reagent for 𝙩𝙚𝙧𝙩-butyl esters synthesis

Aqueous Phosphoric Acid as a Mild Reagent for Deprotection of tert-Butyl Carbamates, Esters, and Ethers

Reagent-free continuous thermal tert-butyl ester deprotection - ScienceDirect

Preparation and Applications of 4-Methoxybenzyl Esters in Organic Synthesis. - Abstract - Europe PMC
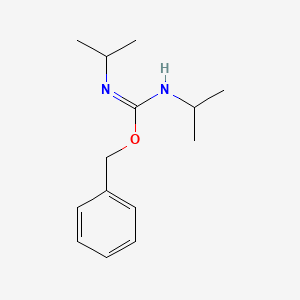
Buy o-Benzyl-n,n'-diisopropylisourea, 2978-10-1
O-tert-Butyl-N,N'-diisopropylisourea 71432-55-8

Advantage of tert-Butyl Esters in organic synthesis & comparison with simple methyl ester.

Efficient and Controllably Selective Preparation of Esters Using Uronium-Based Coupling Agents

ZnEt2 as a Precatalyst for the Addition of Alcohols to Carbodiimides

Alkylating Reagents Containing Phosphoric Ester Groups for Prodrug Design

Enantioselective Total Syntheses of Cyathane Diterpenoids - Nakada - 2014 - The Chemical Record - Wiley Online Library